This page is restricted to SUPER-ED participating hospital sites and serves as a central hub for resources and information related to the Intervention Phase of the study.
🔒 Clinician Access Only
Username: clinician@cup.com
Password: clinician
Note: This page is not accessible to patients or carers.

Investigation of patients presenting with suspected metastatic cancer without an obvious primary site, or malignancy of undefined primary origin (MUO), should be guided by the patient’s clinical presentation. Invasive diagnostic tests may not be clinically appropriate for frail patients who have a poor prognosis or would not be fit for treatment. Clinician discretion is advised. The following recommendations have been adapted from the Australian Optimal Care Pathway for people with Cancer of Unknown Primary and ESMO Clinical Practice Guideline for Cancer of Unknown Primary.
The minimum diagnostic work-up should include:
Additional investigations
Additional investigations are based on the specific presentation and may include:
The CUP Optimal Care Pathway (Full Version) offers detailed guidance on the diagnosis, treatment, and management of CUP.
You can view or download the PDF version here or explore the online version.
Please note: the online version is structured slightly differently and includes a more comprehensive glossary to support understanding.
The CUP OCP Quick Reference Guide is a summarised version that clinicians and other health professionals can quickly refer to.
Cancer of unknown primary | Guide to best cancer care | Cancer Council
Full version of the CUP Optimal Care Pathway for Aboriginal and Torres Strait Islander
Summary of the CUP Optimal Care Pathway for Aboriginal and Torres Strait Islander
The European Society for Medical Oncology (ESMO) is a professional organisation for Medical Oncology. The Clinical Practice Guidelines for Cancer of Unknown Primary (CUP) were established by a panel of multidisciplinary experts across different institutions worldwide, to provide key recommendations for managing CUP patients. The guideline covers diagnosis, risk assessment, classification, treatment and follow-up and incorporate differential diagnostic algorithms to quantifiably distinguish CUP from other cancers.
The ESMO CUP Guidelines:
Cancer of Unknown Primary | ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up

The national multidisciplinary meeting (MDM) of clinical experts in Cancer of Unknown Primary (CUP) is established as part of the SUPER-ED study. The complex nature of CUP, along with the advantages gained from multidisciplinary collaboration, strongly support a dedicated MDM service, as recommended by the CUP Optimal Care Pathway.
Referring clinicians must complete the following forms (Fillable PDF):
Peter Mac Referral form (for patient registration)
SUPER-ED CUP-MDM referral form
Second opinion pathology review request form (if applicable)
Please email completed forms to referrals@petermac.org AND SuperEDstudy@petermac.org. Relevant imaging must be uploaded to Peter Mac PACS.
If the Anatomical Pathology Second Opinion Request form is completed, ensure the pathology report and original stained slides are available for Peter Mac Anatomical Pathology.
Referrals are currently only accepted from participating SUPER-ED sites. The MDM operates during the intervention phase of the study, with each site's start and stop dates defined according to the site randomisation schedule (see Table 1).
Referrals are reviewed on a case-by-case basis, with priority given to patients enrolled in the SUPER-ED study. If the date of the first imaging investigation that detected or suspected metastasis (“DAY 0”) occurs during the intervention phase, the case is prioritised to support faster diagnostic pathways.
The table below includes the dates that SUPER-ED sites will have access to the CUP MDM.
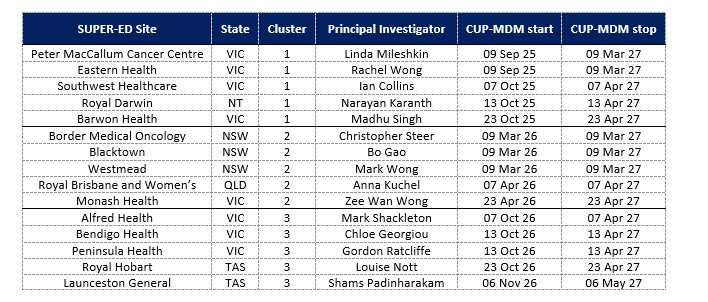
*CUP-MDM start/stop dates in accordance with the intervention phase of SUPER-ED randomisation schedule.
The Virtual CUP-MDM is held fortnightly on Fridays, from 3:00 to 4:00PM via Microsoft Teams. The meeting is facilitated by Peter MacCallum Cancer Centre, with expert input from specialists in nuclear medicine, medical imaging, anatomical pathology, and molecular pathology.
The MDM is particularly valuable for complex diagnostic cases that require multidisciplinary input—such as those involving correlation of clinical history, imaging, histology, and/or molecular results. Potential referrals may be discussed in advance with the CUP-MDM Chair (Prof Linda Mileshkin) or Deputy Chair (Tharani Sivakumaran) to confirm suitability.
The referring clinician, or a delegate, will be required to attend the MDM to present the case. Case discussion notes will be shared to the referring clinician within 4 business days of the meeting; and the patient’s GP (if known).
Due to the confidential nature of patient test results, admission to this meeting is by invitation only. Referring clinicians or their representative, and CUP Care Coordinators from SUPER-ED participating sites are invited to join the meeting. The referring clinician retains responsibility for obtaining patient consent.
The following hospitals have a Picture Archiving and Communication System (PACS) agreement with Peter Mac for the transmission and storage of external imaging and clinical reports:
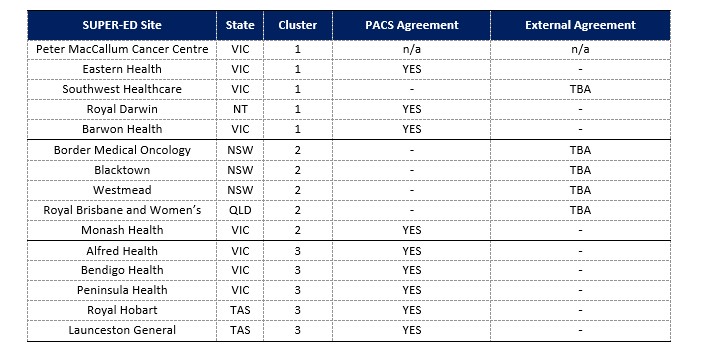
External providers that have a PACS Agreement with Peter Mac:
Breastscreen Victoria; Diagnosticare; Genesiscare, IMED, Lake Imaging; Radiology Tasmania
Virtual CUP MDM Terms of Reference and SOP
Virtual CUP MDM Brief Guidelines for Referring Clinicians
For questions or referrals, contact the MDM Coordinator on +61 3 8559 7453 or SuperEDstudy@petermac.org.
Patients with CUP are often unaware that clinical trials may be an option for their care. Trials can offer access to novel diagnostics, targeted therapies, or alternative standards of care that may not be otherwise available.
As a healthcare professional, identifying suitable trials for CUP patients can be complex due to strict eligibility criteria and the heterogeneity of CUP presentations. However, referring patients to appropriate clinical trials can contribute to improved diagnostic clarity, personalised treatment options, and potentially better outcomes.

We encourage clinicians to consider trial participation as part of the care pathway for CUP patients, particularly if standard diagnostic and treatment approaches are limited.
View Open and Recruiting Clinical Trials for suspected or confirmed CUP.

To support participating hospitals transitioning into the intervention phase, tailored CUP Patient Information Handouts have been developed. These handouts outline site-specific services and resources,including those introduced through the New Model of Care, to ensure patients receive appropriate and coordinated support.
Information Sheets now available for Cluster 1 sites.
CUP Patient Information - Barwon Health
CUP Patient Information - Eastern Health
CUP Patient Information - Peter Mac
CUP Patient Information - Royal Darwin
CUP Patient Information - Southwest Healthcare

Study Coordinator Scope of Practice SOP
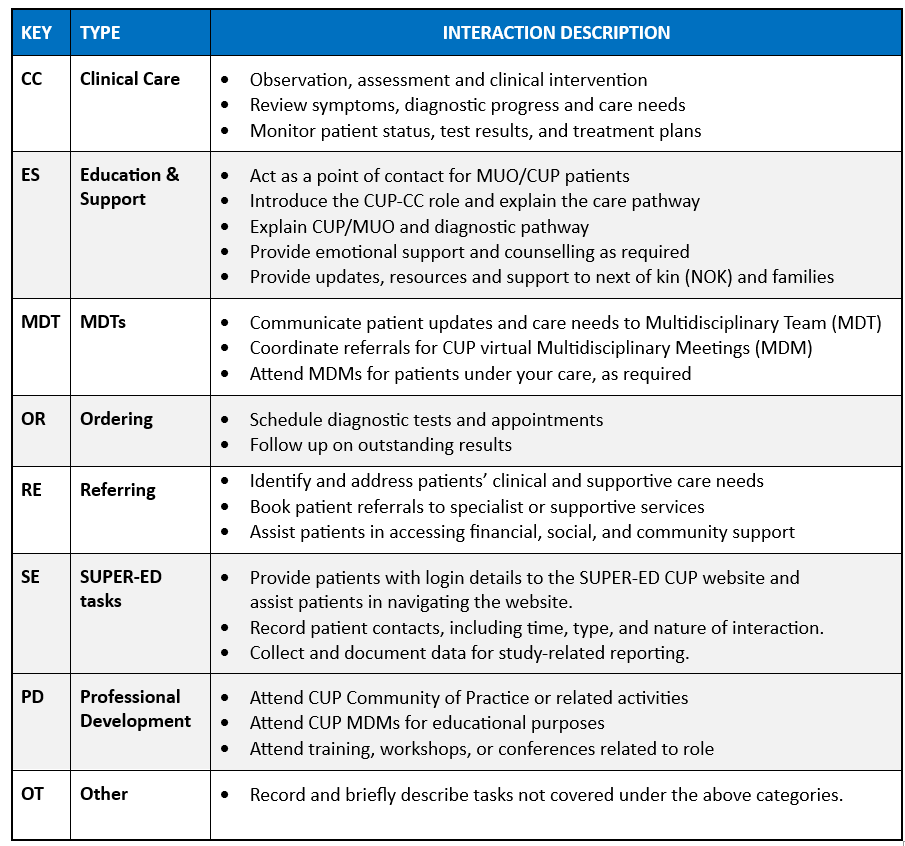
Funded through the SUPER-ED Study, the CUP Care Coordinator (CUP-CC) supports patients with suspected or confirmed Malignancy of Unknown Origin (MUO) or Cancer of Unknown Primary (CUP). The CUP-CC is embedded within existing clinical or non-clinical role/s and is funded for approx. 4 hours per week (0.1 FTE). The CUP Scope of Practice outlines key responsibilities across:
As part of the training package, a curated set of resources has been developed by the CUP Nurse Consultant Lead at Peter MacCallum Cancer Centre. These materials support CUP Care Coordinators in delivering high-quality, patient-centred care for individuals with suspected or confirmed CUP.
Drawing on clinical expertise and practical experience, the resources provide guidance across key areas of CUP management, including:
Find the resources attached below:
Treatment
Cancer of Unknown Primary Treatment
Carcinoma of Unknown Primary Treatment
Symptoms and Side Effects
Wellbeing
Exercise in Cancer Care
Psycho-Social
Communication in Cancer Centre
Adjustment to Cancer Anxiety and Distress
End of Life
Planning the Transition to End-Of-Life Care in Advanced Cancer

The CUP-CC is required to complete the Task Tracker for each patient they engage with during the Intervention Phase of the SUPER-ED Study. This tool captures all activities including (direct/indirect) patient care and the CUP-CC role - helping to quantify workload and inform future planning.

Community of Practice - Terms of Reference
A Community of Practice (CoP) has been established to support the CUP-CC role, recognising the complexity of CUP and the value of peer collaboration. The CoP fosters shared learning, builds workforce capacity, and strengthens care coordination across teams.
Meeting Details
Objectives
Discussion Format

Study Protocol - current approved version
Patient Information and Consent Form
Medicare Patient Consent Form
Master MBS_PBS Consent Form v2.2
Watch and listen to genomic specialists share their expert insights in understanding genomic testing for CUP patients.
The use of molecular profiling tests to identify actionable mutations or determine the tissue of origin is the subject of ongoing research and is not considered part of the standardised diagnostic workup for CUP. However, where this is available (e.g. via research projects or self-funded by patients), it may be helpful and should be considered.
OMICO's Cancer Screening Program provides Comprehensive Genomic Profiling to patients in Australia with advanced, incurable or an earlier diagnosis of a poor prognosis cancer, and refers patients to clinical trials and new targeted therapies where appropriate.
SUPER-NEXT is an observational study assessing the utility of whole genome sequencing for patients with Cancer of Unknown Primary.
Tothill RW, Li J, Mileshkin L, Doig K, Siganakis T, Cowin P., ... Bowtell DD. Massively-parallel sequencing assists the diagnosis and guided treatment of cancers of unknown primary
Journal of Pathology 231(4):413-23. https://doi.org/10.1002/path.4251
Advanced DNA sequencing techniques can help identify the origin of CUP and guide more targeted treatments. In summary, this research highlights the transformative potential of advanced DNA sequencing in managing CUP, offering patients more personalised and effective treatment options.
Researchers analysed tumour samples from 16 patients with CUP using a method called massively-parallel sequencing, which examines the DNA of 701 genes known to be important in cancer. This approach revealed:
· Identification of Cancer Origin: Sequencing helped determine the likely origin of the cancer in several cases, providing crucial information for treatment planning.
· Potential Treatment Targets: The analysis identified specific genetic alterations that could be targeted with existing or experimental therapies.
· Improved Diagnostic Accuracy: The study highlighted the limitations of traditional diagnostic methods and the added value of advanced sequencing in challenging cases.
For patients with CUP, this study offers several benefits:
· Clearer Diagnosis: Advanced sequencing can provide a more accurate identification of the cancer's origin, leading to more informed treatment decision.
· Personalised Treatment Options: Identifying specific genetic alterations allows for the consideration of targeted therapies, which may be more effective than standard treatments.
· Access to Clinical Trials: Patients may become eligible for clinical trials testing new therapies tailored to their genetic profile.
CUP is a complex condition where the cancer's origin is unknown, making treatment challenging. This study underscores the potential of advanced DNA sequencing to:
· Enhance Diagnostic Accuracy: Providing clearer insights into the cancer's origin can lead to more precise diagnoses.
· Guide Targeted Therapies: Understanding genetic alterations enables the use of treatments tailored to the individual's cancer profile.
· Improve Patient Outcomes: By moving beyond traditional treatments, patients may
experience better responses and outcomes.
Posner A, Prall OWJ, Sivakumaran T, Etemadamoghadam D, Thio N, Pattison A., ...Tothill RW.
A comparison of DNA sequencing and gene-expression profiling to assist tissue of origin diagnosis in cancer of unknown primary Journal of Pathology PMID: 36287571. https://doi.org/10.1002/path.6022
Advanced DNA and RNA tests can help identify the origin of cancers of CUP in about one-third of cases, providing more targeted treatment options.
This study compared two advanced tests - DNA sequencing and gene expression profiling (GEP) - to determine the origin of CUP in215 patients. Key findings include:
Overall, DNA sequencing was more effective than GEP in providing diagnostic clarity.
For patients with CUP, these advanced tests offer:
CUP is challenging to treat due to its unclear origin. This study highlights that:
In summary, advanced DNA and RNA tests play a crucial role in diagnosing and treating CUP, offering patients more precise and effective care options In Brief
Advanced DNA and RNA tests can help identify the origin of cancers of CUP in about one-third of cases, providing more targeted treatment options.
Kato S, Krishnamurthy N, Banks KC, De P, Williams K, Williams C., ... Kurzrock R. Utility of Genomic Analysis In Circulating Tumor DNA from Patients with Carcinoma of Unknown Primary
©2017 American Association for Cancer Research. 2017 Aug 15;77(16):4238-4246. https://doi.org/10.1158/0008-5472.can-17-0628
Using a simple blood test called a liquid biopsy,researchers found that most patients with CUP have unique, drug-targetable genetic changes, offering new hope for tailored treatment.
In this study, researchers analysed blood samples (circulating tumour DNA or ctDNA) from 442 people diagnosed with CUP. They used advanced genetic testing to look at 54–70 cancer-related genes. The results showed:
· 80% of patients had detectable ctDNA alterations.
· 66% (290 out of 442) had at least one clear,actionable genetic change (excluding uncertain variants).
· Among those with characterized alterations,nearly 99.7% had at least one genetic change that could, in theory, be targeted with existing or investigational treatments.
This study demonstrates that:
· A non-invasive blood test can detect genetic changes in most people with CUP.
· Almost every patient with such changes - 99.7% -could potentially benefit from a targeted therapy or immunotherapy, based on their specific alterations.
· These findings support the use of liquid biopsies in clinical trials and future treatment pathways tailored to a person’s unique cancer biology.
CUP is traditionally hard to treat because doctors don’t know where the cancer started, so treatment is often limited to standard chemotherapy. This study shows that:
· Most CUP patients have actionable genetic changes that could guide more precise, personalised treatments.
· A simple blood test (liquid biopsy) makes testing easier - especially when tissue samples are limited or hard to obtain.
· Liquid biopsies may open a path toward more effective, targeted care for many patients with CUP.
Ross JS, Sokol ES, Moch H, Mileshkin L, Baciarello G, Losa F., ...Krämer A. Comprehensive Genomic Profiling of Carcinoma of Unknown Primary Origin: Retrospective Molecular Classification Considering the CUPISCO Study Design. Oncologist. 26(3) https://doi.org/10.1002/onco.13597
In a recent study reviewing genomic data from people with CUP, one-third of patients would have been eligible for personalized treatment in the ongoing CUPISCO trial - highlighting how genetic profiling may open new therapeutic avenues.
CUP is traditionally treated with broad chemotherapy due to unclear tumor origins and limited treatment options. This study demonstrates that genomic profiling can meaningfully broaden access to targeted therapies or immunotherapy, offering hope for more effective care in a significant subset of patients. It supports the growing move towards personalised cancer treatment even when the primary site isn't identified.
Richard J. Rebello, Atara Posner, Ruining Dong, Owen W. J. Prall, Tharani Sivakumaran, Camilla B.Mitchell., ... Richard W. Tothill. Whole Genome Sequencing to improve the tissue of origin diagnosis and treatment options for patients with Cancer of Unknown Primary
Nature Communications, 16, Article 4422 . https://doi.org/10.1038/s41467-025-59661-x
A new study shows that whole-genome and transcriptome sequencing (WGTS) can give people with CUP clearer answers about where their cancer began and open up more treatment options, including access to clinical trials. This advanced testing offers new hope by replacing uncertainty with more personalised choices.
Researchers studied 72 CUP patients, comparing WGTS with standard genetic panel tests. They found WGTS:
· Detected all the same changes as panel tests plus more, adding useful information.
· Predicted where the cancer started in 71% of patients (vs. 34% with panel tests).
· Identified potential treatment targets in 74% of patients, increasing therapy and trial options.
WGTS can:
· Provide clearer answers about the cancer’s origin.
· Guide more personalised treatments such as targeted therapy or immunotherapy.
· Improve access to clinical trials.
· Sometimes use a blood test when tissue samples are limited.
CUP is challenging because treatment is less precise without knowing where the cancer began. WGTS offers a powerful new tool that:
· Improves diagnostic certainty
· Expands treatment options
· Brings greater hope to people facing CUP.
Arielle van Mourik, Gina Tonkin-Hill, John O'Farrell, Shohei Waller, Lavinia Tan, Richard W Tothill., ... Linda Mileshkin. Six-year experience of Australia’s first dedicated CUP clinic British Journal of Cancer 129:301–308. https://doi.org/10.1038/s41416-023-02254-6
Australia’s first dedicated Cancer of Unknown Primary (CUP) clinic is helping patients by providing clearer diagnoses, better access to DNA testing, and more personalised treatments. The study shows that patients seen in the clinic had improved options, including targeted therapies and clinical trials, leading to better outcomes and greater hope.
This study looked at the first six years of Australia’s CUP clinic at Peter MacCallum Cancer Centre. CUP is a rare diagnosis where cancer has spread, but doctors cannot find where it started. This makes treatment decisions more difficult.
The clinic supported patients by:
· Confirming whether they truly had CUP, another cancer, or a non-cancer condition.
· Completing important tests, including DNA (genomic) testing.
· Linking patients to clinical trials and newer treatments.
Patients benefited through:
· Clearer diagnosis, avoiding unnecessary or incorrect treatments.
· Better access to DNA testing, giving treatment clues in about one-third of patients.
· More personalised treatment, such as targeted therapies or immunotherapy, which were linked to longer survival compared to standard chemotherapy.
· Access to clinical trials, offering promising treatments not otherwise available.
For people with CUP, having a dedicated clinic means care is more focused, personalised, and hopeful. By combining thorough diagnosis, modern DNA testing, and access to new treatments, the CUP clinic improves the chances of finding effective therapies and supporting patients through a challenging diagnosis.
Here are the links to other clinician resources. You may also use the quick links on the right side of the page to navigate.

Investigations for a suspected cancer can lead to various diagnostic outcomes. To learn more about the different types of diagnosis, click here.




